Jesko Koehnke, Andrew Bent, Greg Mann
Cyclic peptides have great potential as novel pharmaceuticals but to date their production has proven challenging for synthetic chemists. Marine cyanobacteria are known to produce a diverse family of cyclic peptides, the cyanobactins, through the ribosomal synthesis of precursor peptides and post-translational tailoring. The patellamides, a member of the cyanobactin family, are cyclic octapeptides containing D-stereo centres and heterocyclised amino acids. The patellamides are derived from a precursor peptide, PatE, which undergoes several modifications, catalysed by the enzymes PatA, B, C, D, F and G, to form the final cyclic peptide (macrocycle). These modifications include heterocyclisation, proteolysis, oxidation and macrocyclisation.
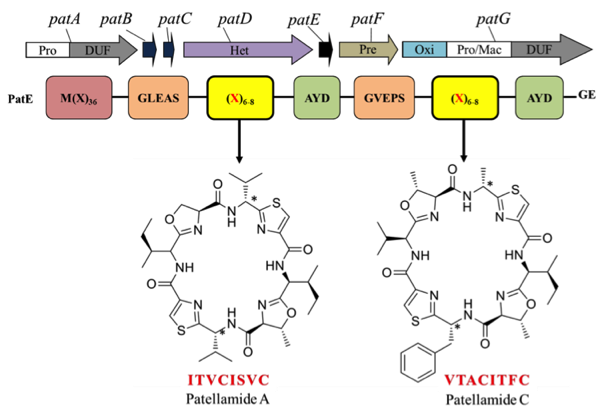
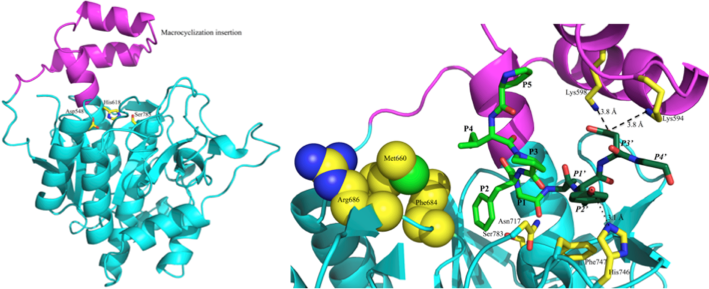
We have explored structurally and biochemically the individual enzymes of the patellamide pathway in order to characterise and potentially adapt their functions for use in the production of novel cyclic peptides. To date we have solved the structures of the PatG macrocyclase domain and its complex with a substrate mimic, highlighting the key interactions which enable macrocyclisation over standard proteolysis (Figure 2). We have also solved the structure of the PatA protease domain.
J. Koehnke, A. Bent, W.E. Houssen, D. Zollman, F. Morawitz, S. Shirran, J. Vendome, A.F. Nneoyiegbe, L. Trembleau, C.H. Botting, M.C.M. Smith, M. Jaspars & J. H Naismith, The mechanism of patellamide macrocyclization revealed by the characterization of the PatG macrocyclase domain, Nature Structural & Molecular Biology, advance online publication, doi: 10.1038/nsmb.2340 (2012).

