Christos Pliotas, Richard Ward, Emma Branigan
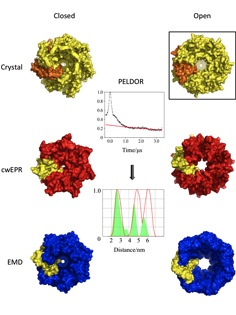
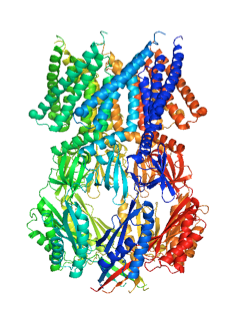
The heptameric mechanosensitive channel of small conductance (MscS) is vital for cell survival as it opens in response to direct lipid bilayer tension to relieve turgor pressure and prevent cell lysis. MscS is a widely used model system for membrane protein ion channels and constitutes a potential drug target. Upon opening, the 15 Å wide and non-selective pore created, allows passage of ion and solutes through the cell membrane resulting in a total conductivity of 1nS. The basis of gating has been studied by protein X-ray crystallography, cwEPR spectroscopy and extrapolated motion dynamics. The models of gating derived by these techniques are mutually incompatible and each reports very different arrangements of the transmembrane helices.
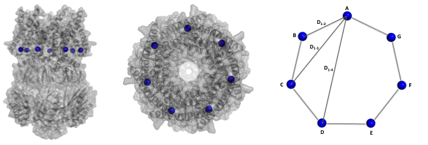
Our approach for channel gating investigation involves the attachment of spin labels to site-directed mutants on key structural elements of MscS. We subsequently apply pulsed electron-electron double resonance (PELDOR) spectroscopy (also known as DEER) on the selected mutants, which allows the accurate measurement of distances within transmembrane regions of the heptamer. These distances from multiple mutants provide an orthogonal and independent assessment of the validity of the competing structural models while they offer the advantage of measurements while MscS is in solution, under physiological conditions. In addition, membrane protein X-Ray crystallography is applied in parallel to PELDOR in order to further verify the validity of the resulting distance distributions.
Pliotas C, Ward R, Branigan E, Rasmussen A, Hagelueken G, Huang H, Black SS, Booth IR, Schiemann O, Naismith JH. (2012) Conformational state of the MscS mechanosensitive channel in solution revealed by pulsed electron – electron double resonance ( PELDOR ) spectroscopy. PNAS, 109:E2675-82.

