Recently the crystal structure of the Ubiquitin loaded E2 enzyme (UbcH5a) in complex with the RING domain dimer of the E3 ligase RNF4 was solved (Fig. 1).1 The E2~Ubiquitin conjugate was stabilised by introducing lysine to the E2 active site thus replacing the thio-ester linkage between E2 and Ubiquitin with an isopeptide linkage.
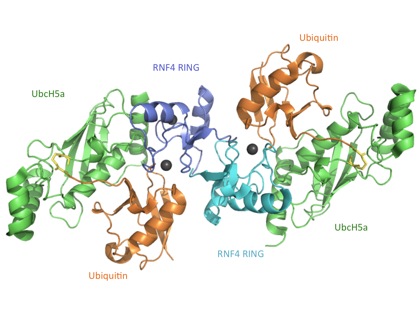
Fig. 1: Crystal structure of E2 UbcH5a (green) conjugated to Ubiquitin (orange) in complex with the RING domain dimer of RNF4 (blue and cyan). Zinc ions are shown as grey spheres and the isopeptide linkage is highlighted in yellow.
1. Plechanovová, A., Jaffray, E.G., Tatham, M.H., Naismith, J.H., and Hay, R.T. (2012). Structure of a RING E3 ligase and ubiquitin-loaded E2 primed for catalysis. Nature 489, 115-120.

